Cl charge element12/10/2023 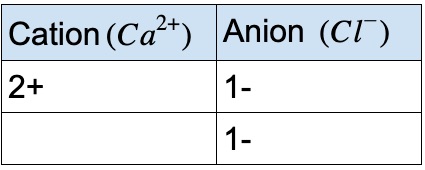
In conclusion, it is very clear now the charge of zinc is 2 plus. Remember there are a few special transition metals like zinc we have mentioned before only have one charge. Once we reach group 4a, we can start back down, followed by negative 3, negative 2, negative 1 and end up with 0 for group 8a. Then group 3a has plus 3 charges, and next to it, group 4a has a plus or minus 4 ionic charge. Skipping the transition metals which can have more than one ionic charge. Group one has plus one charge, in group two, has plus two charges. How to find the charge on the periodic table So zinc is metal and will lose electrons to form the zinc ion. We need two chlorine ions to balance out the two plus of zinc, so we have zinc chloride (ZnCl2). For example, zinc can lose 2 ionic to something like chlorine (Cl). If you are curious about where it loses these two electrons, in general, it loses them to another element when it forms a bond. Unlike most of the transition metals, zinc is always 2 plus with 2 plus ionic. However, zinc (+2), cadmium (+2), and silver(+1) are special exceptions here. We have to look and see what it is bonded to. We really don’t know what the ionic charge of these transition metals. Many transition metals often have more than one charge. When zinc loses all the electrons on its fourth shell, it turns out to be a zinc ion(Zn+2), a positively charged cation. Since electrons are negative, zinc loses 2 negative charges, which makes the charge of zinc becomes two plus. Zinc is likely to lose the two electrons in its fourth subshell. The number of protons and electrons is equal, so it doesn’t have a charge, you can also say the charge is zero. The atomic number 30 means the number of protons in it. On the chemical periodic table of elements, zinc is the 30 elements. 
That means it still has to have eight protons and ten electrons. 
But if we have an oxygen atom with 2 minus on it, in this case, it has an overall charge of negative 2. So it is going to have eight electrons to make it neutral. The number eight means no matter what it has to have eight protons. For example, if we have an oxygen atom, which is atomic number eight. In chemistry, the charge can mean that the number of protons is not equal to the number of electrons. On the contrary, an atom is negative, if it takes on any electrons in a reaction, or if electrons deflected toward itself. How many electrons exhibit such a tendency, means how much positive charge it will have. When an atom loses electrons or electrons deviate from itself, it will have a positive charge. Because the contrary charge pulls towards the protons. They zip around protons at lightning speed. They orbit the nucleus, but not like an orbit with the planets. Neutrons are neutral, which means they are not charged, and have almost equal large mass to protons, also in the nucleus.Įlectrons are negatively charged and have a tiny mass. Protons are positively charged, have large relative mass, and make up the nucleus. Everything is made up of atoms, and atoms are composed of three kinds of tiny particles: protons, neutrons, and electrons. In this way, you will have a more clear mind. įirst of all, let’s figure out some basic chemistry concepts. We will provide easily digestible examples and explanations on this topic. From these simple but important questions, let’s review some interesting chemical knowledge together. What is the charge of zinc is a basic question in high school chemistry class.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
